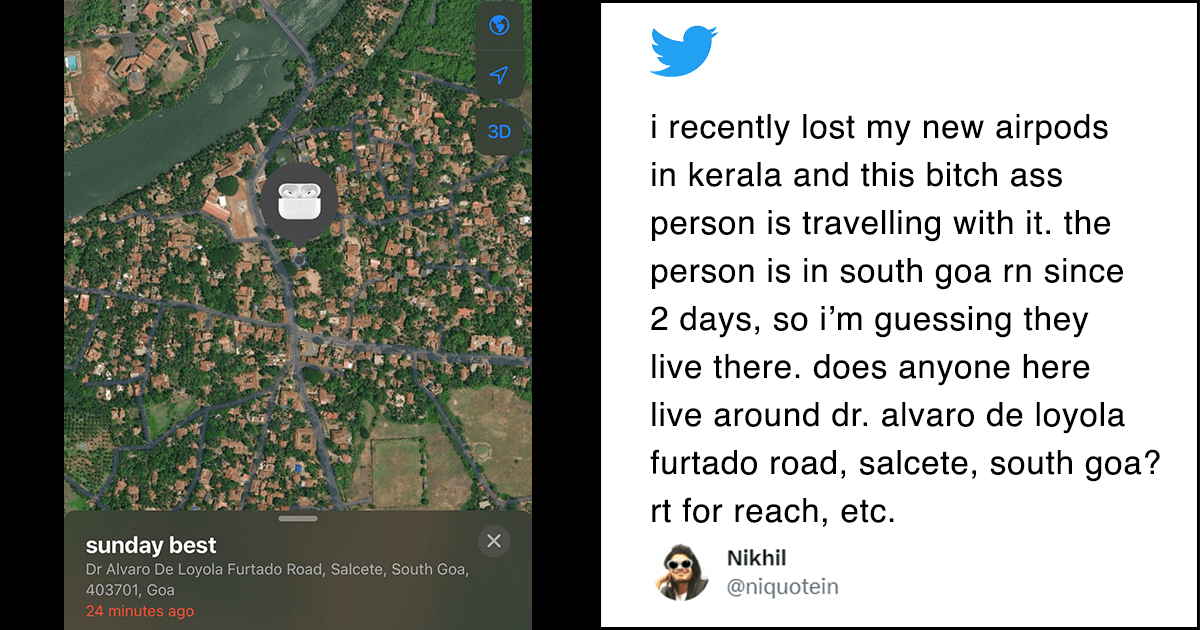
Scientists, including one of Indian origin, have developed a button-sized, garnet-based rechargeable battery that may power vehicles, electronics and grids for storing renewable energy.
The new battery developed by Venkataraman Thangadurai and colleagues at the University of Calgary in Canada is chemically stable, non-flammable and can operate safely at a higher voltage than existing batteries.
Electric vehicles promise to revolutionise transportation but they need safer, better-performing batteries. Current lithium-ion batteries have several issues, including leakage, poor chemical stability, flammability and limited operating voltage or energy density.
“The technology we have developed would enable absolutely stable, robust, safe, high-powered, all solid-state lithium batteries for future energy storage,” said Thangadurai.

Such next-generation batteries have many potential applications, including in electric vehicles, consumer electronics, solid-state gas sensors and electrical grids for storing power generated by renewable energy and providing electricity during peak demand times.
Existing lithium-ion batteries used in electric and plug-in hybrid vehicles, as well as in portable electronics, use membranes of organic polymer compounds and lithium salts as the electrolyte.
The electrolyte in a battery separates the two electrodes (the positive cathode and the negative anode) and conducts the lithium ions between the electrodes during charging and discharging cycles. Currently used organic polymer-based electrolytes are flammable, so fire is a safety issue.
Instead of organic polymers for their battery, researchers used a solid ceramic electrolyte, which does not burn.
The research team also used, for the first time, a technique called atomic layer deposition to place a thin film of aluminum oxide on top of a garnet structure coating the ceramic electrolyte.
With their chemically engineered, garnet-based, ceramic electrolyte, the team showed there was “negligible interface resistance” between the lithium metal anode and the ceramic electrolyte interface. This resulted in fast transport of charges and higher performance overall.
Their newly developed interface chemistry also enabled the team to use a lithium metal anode and a high-voltage cathode, which will significantly boost their battery’s energy storage capacity and operating voltage compared with existing lithium-ion batteries.
While other research groups in the world have used garnet to build lithium batteries, “we showed we can use the lithium metal very efficiently, with the lowest interface-charge transfer resistance between the lithium electrode and the garnet electrolyte,” said Thangadurai.