While we all are waiting for the government to end this lockdown and let us out of our houses, the scientists and doctors are doing their best to develop vaccines as a cure to let us all out get back to our ‘normal’.

As of now, there are close to 70 vaccines that are being worked on across the world. As the vaccine might take more than 15 months to be used on the Covid-19 affected patients, doctors are looking for alternates that could make the infected survive.
The one vaccine that has got the most votes for is anti-malarial drug but a recent study has shown that the antiviral remdesivir, an experimental Ebola drug, may stop Sars-CoV-2, which causes Covid-19, from replicating and thus making it harmless.
The drug is developed by a US-based biotechnology company, Gilead Sciences Inc., which has a patent on the drug in India. Remdesivir is among four treatments being assessed under the World Health Organization’s unanimity trial to find an effective treatment for Covid 19.
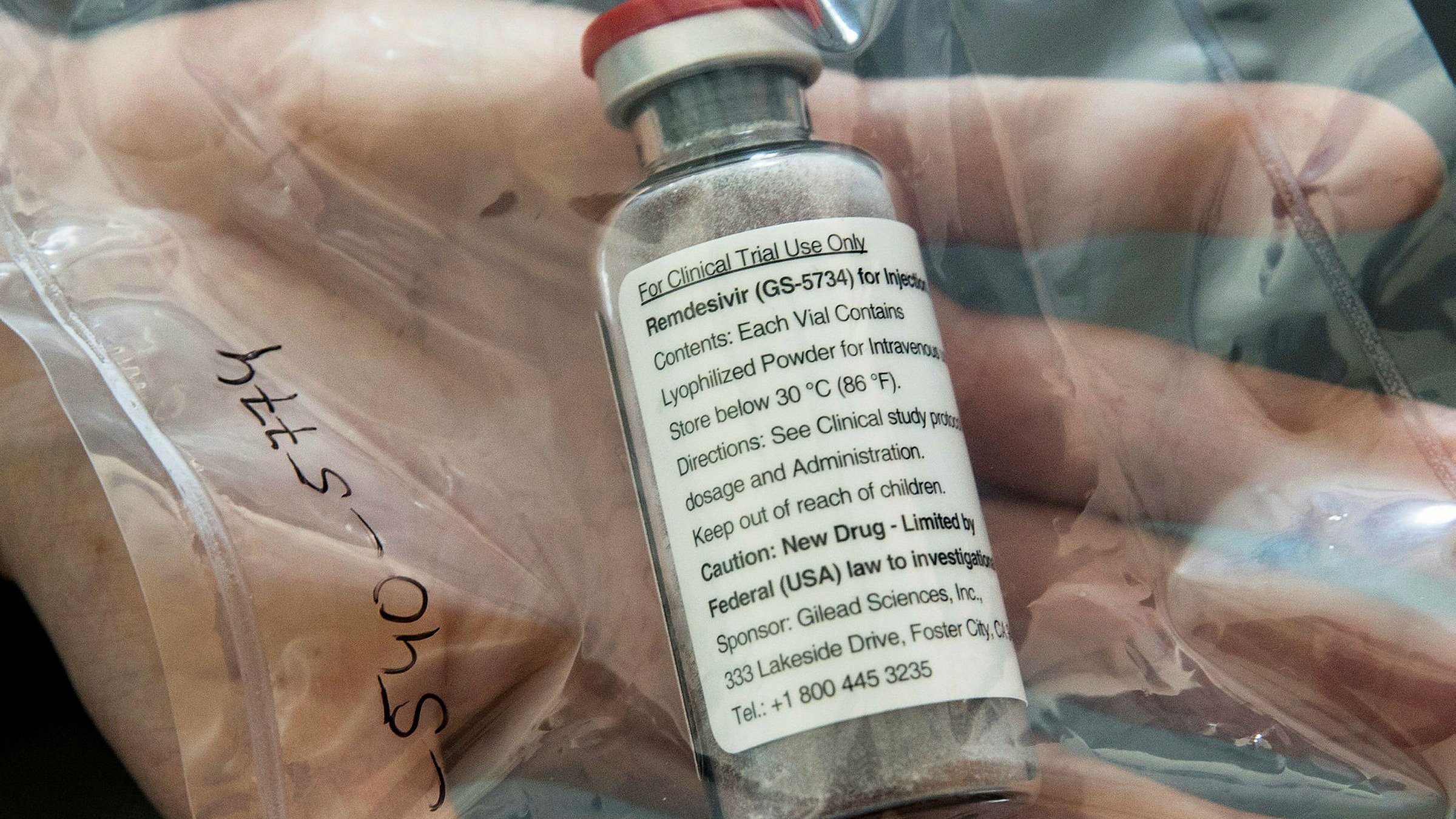
India is participating in the trial. Gilead has also started two Phase 3 clinical trials on the safety and efficacy of the drug. Phase 3 trials are required by the US Food and Drug Administration and other regulators to approve the drug.
According to a research paper in the Biological Chemistry Journal published on Monday, remdesivir can inhibit coronavirus polymerases, enzymes that bring about the formation of polymers, particularly DNA and RNA.
The polymerase of Sars-CoV-2 is like the engine of the virus and remdesivir tricks the virus by mimicking its building blocks. These coronavirus polymerases are sloppy and they get fooled, so the inhibitor gets incorporated many times and the virus can no longer replicate.

The drug was first used on a 35-year old who returned to the US after a family visit to the Chinese city of Wuhan, with a four-day cough and subjective fever, who was found to have improved after receiving remdesivir.
The man developed pneumonia and recorded an oxygen saturation of 90%. Treatment with intravenous remdesivir started on the evening of day 7. On day 8, his condition is said to have improved and his oxygen saturation values improved to 94-96%.

In another paper published on April 10, the journal documented the cases of 53 patients with severe symptoms of Covid-19 who were treated with the drug and clinical improvement was seen in 36 of them.
However, only placebo-controlled trials have provided an accurate picture. Remdesivir has been previously tested for treatment of Ebola.

It has provided promising results in animal studies for Middle East Respiratory Syndrome (Mers-CoV) and severe acute respiratory syndrome (Sars), which are also caused by coronaviruses, suggesting it may have some effect in patients with COVID-19.
Remdesivir was used in Ebola. And in a Covid-19 observational study it was found to benefit 2 in 3 patients in terms of critical patients going off ventilator and not needing oxygen support.

One of many questions that most of the studies currently aim to answer is whether the treatment duration can be shortened from 10 days to 5 days. And because the Gilead Sciences Inc. holds the patent, the government needs to take various steps to make the drug reach India for the treatment.