As the coronavirus has raged on, desperate research into ways to cure it has been going at at a frantic pace. While there’s still no sureshot vaccine or immediate cure, there are certain medicines slowly being developed that can help treat the dreaded illness. Now, there’s one being made in India.
Mumbai based Glenmark Pharmaceuticals has made the antiviral drug FabiFlu, which has been approved for the treatment of mild to moderate COVID-19 patients.
This makes Glenmark the first Indian company to commercially produce an antiviral drug for the treatment of moderate coronavirus. They received marketing and manufacturing approval from the Indian drug regulator, Drug Controller General of India (DCGI) and launched today.
FabiFlu the first oral Favipiravir-approved medication in India for the treatment of COVID-19 https://t.co/XWpxp7IlA3
— The Hindu (@the_hindu) June 20, 2020
FabiFlu costs Rs 3500 for 34 tablets, which comes out to Rs 103 per tablet.
The dosage is 200 mg X 9 tablets on day one and 200 mg X 4 tablets a day for 14 days.

Clinical trials of mild to moderate patients found an efficacy of 80% in those treated.
Glenmark conducted clinical trials among 90 mild and 60 moderate COVID-19 patients across 11 sites in India.
The drug essentially works by getting into cells and inhibiting viral replication to reduce the viral load. A high rate of viral replication can be controlled with early use of antiviral drugs.
It is important to use these in the early stages, as viral replication in the later stages slows down and the body’s violent immune response drives disease to complications and organ failure.
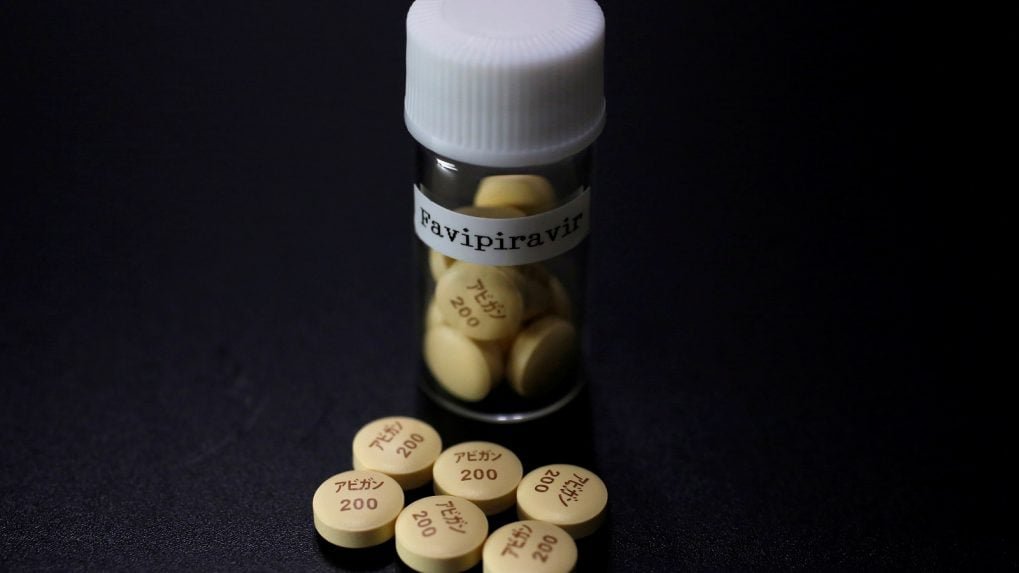
The drug Favipiravir is currently sold under different brand names in certain countries of the world to treat coronavirus. With the arrival of it in India, let’s hope our patients can be given some relief.