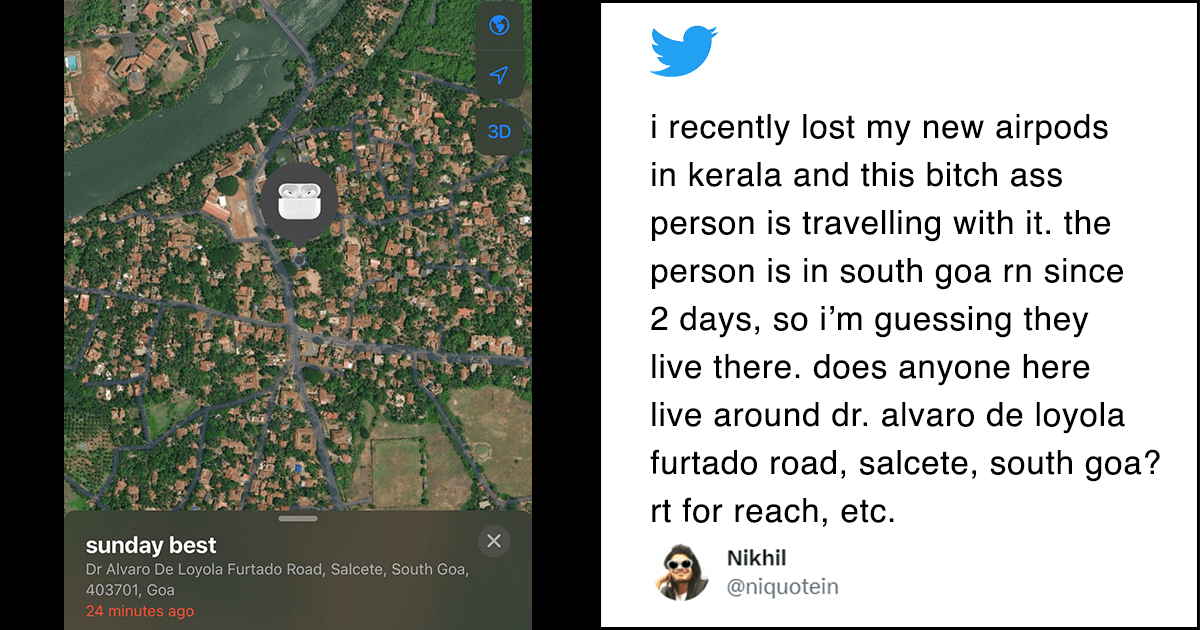
Lockdowns, self isolation, social distancing, these are all terms that are used as temporary measures to control the spread of Covid-19. These measures are to be used until scientists and doctors don’t find a cure.
What is that cure? Nobody knows.
While it might take another 18-24 months for a Coronavirus vaccine to be ready, doctors are trying to figure out an alternate to save their patients.
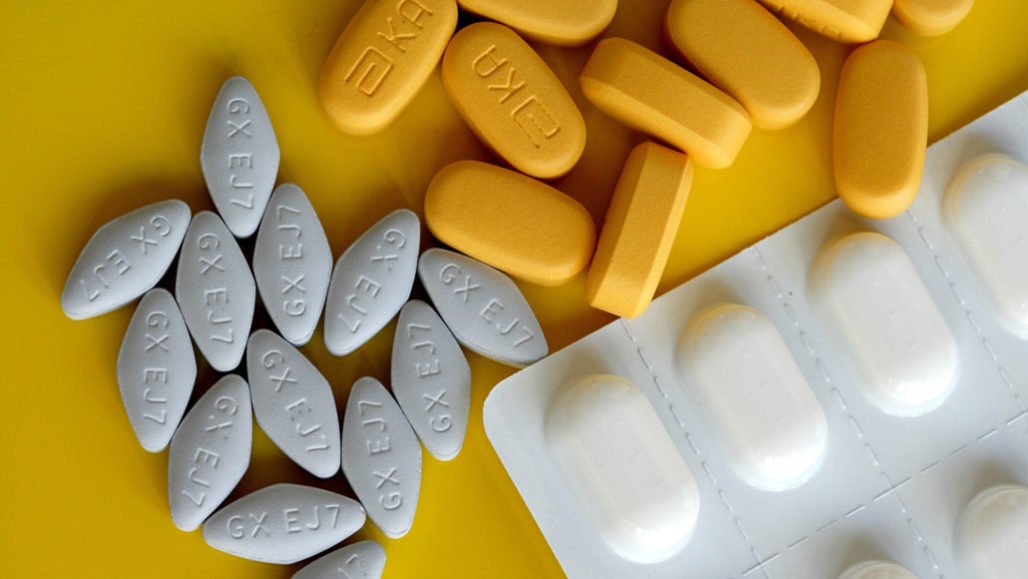
One such alternate with effective measures is the drug that is not not just cheap, safe but also widely available. The drug used to treat malaria.
Hydroxychloroquine, the drug used to treat malaria has emerged as the most sought-after medicine in the world after preliminary trials from China said it boosted recovery and lowered the severity of Covid-19, which has sickened 1.2 million and killed 65,000 around the world within four months.
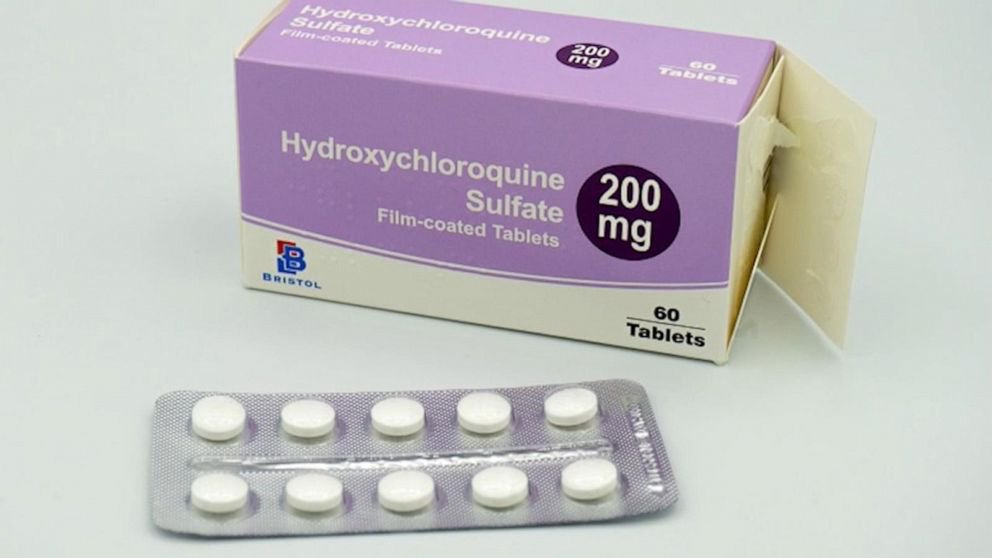
India, thankfully, is the biggest producer of this drug and since its increased demand globally, India has banned the export of the drug and its active pharmaceutical ingredients (APIs), which are the chemicals used to manufacture the drug.
What led to this discovery and global demand for hydroxychloroquine were the results of a small placebo-controlled clinical trial in China that revealed the anti-malarial drug not only shortened the duration but also reduced the severity of cough, fever and pneumonia in patients with mild and moderate disease.
The drug also prevented the illness from worsening in patients as compared to the placebo group. A placebo-controlled trial is done with two groups of patients, one of which is given a placebo with no drug action, while the drug is tested on another group.
This method rules out bias in the outcome and ensures the drug’s effects are actually caused by the treatment and not external factors.
Since the Sars-CoV-2, the virus that causes Covid-19, is about four months old, clinicians worldwide have been depending on experimental therapies using existing drugs in different permutations and combinations to treat symptoms and stop disease progression.
Repurposing medicines with established safety and drug action on the human body is the fastest way to treat the new diseases against which there is no clinically proven treatment.
It allows clinicians and public health specialists to begin treatment until drugs and vaccines with specific action against Covid-19 are approved.
A recent paper published in the Annals of Internal Medicine stated that data to support the use of hydroxychloroquine and chloroquine are limited and inconclusive and there was an urgent need for well-done, randomised clinical trials to test potential therapies, including hydroxychloroquine.

India has approved the combination of two antiviral drugs used to treat HIV, lopinavir and ritonavir (400 mg and 100 mg, respectively), to treat Covid-19, but the first major study of Covid-19 treatment published in the New England Journal of Medicine (NEJM) found it did not work on severely ill patients in China.
Based on a controlled trial on 199 hospitalised patients in China, it stated that the treatment with lopinavir-ritonavir was not associated with a difference from standard care in the time to clinical improvement.
Globally, there are 332 registered clinical trials, of which 188 are open for recruitment and 146 trials are preparing to recruit, most of these trials are taking place in China, South Korea, and high-income countries in Europe and North America.
But until we have a specific drug, therapy or vaccine against Covid-19, drugs like hydroxychloroquine are the best bets to treat and protect patients.